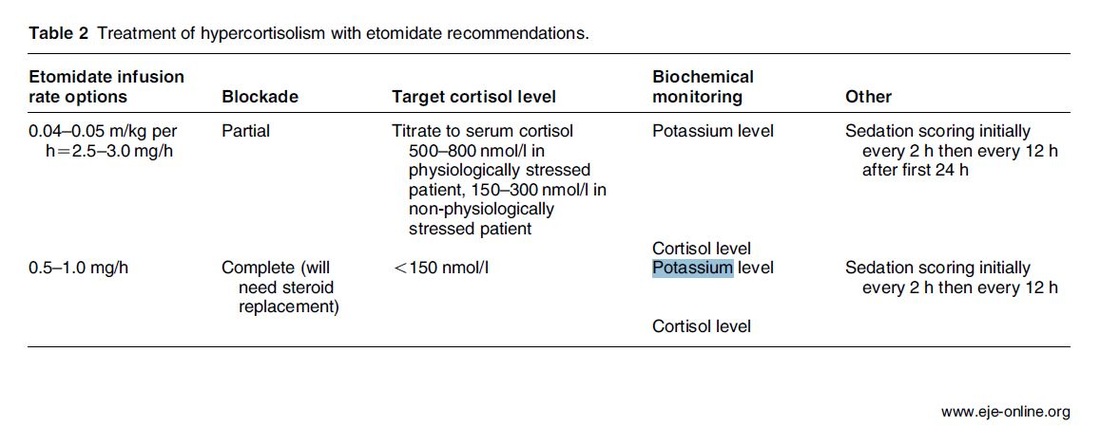
The Problem An endogenous Cushing's syndrome, mostly caused by an adenoma of the pituitary gland, is associated with significant morbidity and mortality when left untreated. The condition is closely associated to life-threatening infections, diabetes mellitus, hypertension and increased risk associated with surgery. For Cushing's disease the first line therapy is surgical removal of the pituitary tumor. Sometimes though urgent medical therapy is needed first. It has been shown, that surgical risk may be significantly reduced if cortisol concentrations are normalised preoperatively. Conditions requiring urgent cortisol-lowering measures are severe biochemical disturbances (e.g. hypokalaemia), immunosuppression or mental instability. Medical Treatment OptionsKetokonazole (yes, the antifungal agent) and metyrapone are used to suppress adrenal steroidogenesis at enzymatic sites. Both agents carry the risk of postential side effects. Mifepristone, a glucocorticoid receptor antagonist, and pasireotide, a new targeted pituitary therapy, are alternative agents. However, they also have their limits and side effects. EtomidateNow that's where etomidate joins the game. Interestingly, etomidate and ketokonazole are chemically closely related... they are both members of the imidazole family. Etomidate is used as an anaesthetic agent since 1972 and became popular for hemodynamic stability and the lack if histamine release. In 1983 a Lancet article noted an increased mortality when etomidate was used in critically unwell patients. In 1984 an article in Anaesthesia first showed a link to low serum cortisol levels caused by etomidate. Until now the discussion continues, whether a single induction dose actually negatively influences patient outcome. A meta-analysis in 2010 was unable confirm this apprehension and the debate continues. Fact isEtomidate suppresses the production of cortisol by inhibiting the mitochondrial cytochrome p450-dependent adrenal enzyme 11-beta-hydroxylase and therefore lower serum cortisol levels within 12 hours. In higher doses it also blocks side chain cleavage enzymes and also aldosterone synthase. It might even have anti-proliferative effects on adrenal cortical cells. On this basis the idea arose, that etomidate might be a useful therapy for severe hypercortisolaemia. Continuous Etomidate - What's the EvidenceA review article by Preda et al. in 2012 identified 18 publications about the primary therapeutic usage of etomidate in Cushing's syndrome, most of which were case reports. Review of current literature reveals that etomidate indeed suppresses hypercortisolaemia safely and efficiently in patients requiring parenteral therapy. Moreover, etomidate shows a dose-dependent suppression and allows adjustment of the medication to target cortisol levels. At recommended dosages etomidate is considered safe with almost no serious side effects. The authors conclude, that etomidate is a useful therapeutic option in a hospital setting when oral therapy is not tolerated or inappropriate. Take home- Continuous etomidate (in non-hypnotic doses) reduces cortisol concentrations in a dose-dependent manner in both hyper- and eucortisolaemic subjects
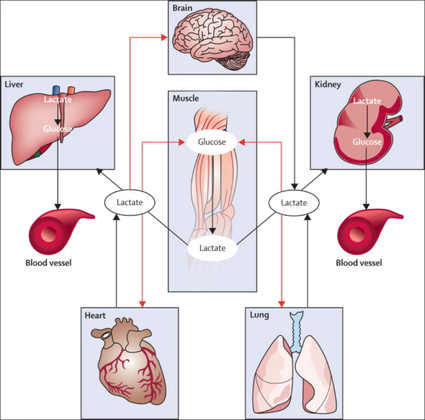
- The application of continuous etomidate in Cushing's disease is safe and efficient - After termination of infusion adrenocortical suppression persists for about 3 hours - The suspicion, that a single dose of etomidate for rapid sequence inductions might negatively influence patient outcome in the critically ill remains a matter of debate J Clin Endocrinol Metab. 1990 May;70(5):1426-30. Preda et al. European Journal of Endocrinology (2012) 167 137-143 OPEN ACCESS Soh et al. Letter to the Editor, European Journal of Endocrinology (2012) 167 727–728 Ge et al. Critical Care201317:R20 OPEN ACCESS  The discussion on the so-called lactic acidosis and its causes have become increasingly attractive over the last couple of years as several biochemical explanations are challenged. A significant confusion persists on the various relationships between lactate, lactic acid and metabolic acidosis. Most clinicians continue to refer to the traditional understanding of impaired tissue oxygenation causing increased lactate production, impaired lactate clearance and therefore resultant metabolic acidosis. Just recently we had a discussion on our ward round on this topic when a team member presented the most recent article of UpToDate online on the causes of lactic acidosis. The authors state that 'Lactic acidosis is the most common cause of metabolic acidosis in hospitalised patients' and that 'Lactic acidosis occurs when lactate production exceeds lactate clearance. The increase in lactate production is usually caused by impaired tissue oxygenation...'... finally suggesting that lactate is no good! These statements support the classical understanding that: - Hyperlactatemia is caused by tissue hypoxemia, and - This in turn then leads to a metabolic acidosis called lactic acidosis This biochemical understanding has persisted for decades, but there are some good reasons to vigorously challenge this traditional aspect on the 'bad' lactate. Lactate turns out to be by far more complex in its characteristics and functions, so I decided to try and make a short but comprehensive overview of this molecule. What is lactate?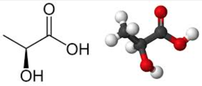 Lactate is a small organic molecule with the chemical formula CH3CH(OH)CO2H and structurally looks like on the image to the left. It is produced in the cytoplasm of human cells mainly by anaerobic glycolysis by the conversion of pyruvate to lactate by LDH. This chemical reaction results typically in a blood lactate to pyruvate ratio of about 10:1. And while lactate is produced, NAD+ also is incurred, and this actually can accept protons itself, so does not result in acidosis itself. Lactate arises from the production of energy by consuming glycogen and glucose.
|
|||||||
| Click here to read | |
| File Size: | 164 kb |
| File Type: | |
In order to answer this question the investigators assigned organ donors after declaration of death according to neurologic criteria into two groups. They were either treated with mild hypothermia (34 to 35°C) or with normothermia (36.5 to 37.5°C). The target temperature was maintained until the patients were transferred to theatre for transplantation.
Primary outcome of this trial was delayed graft function among recipients. Secondary outcomes included the rates of individual organs transplanted into each treatment group at the total number of organs transplanted from each donor.
This trial had to be stopped early as an interim analysis showed significant efficacy of mild hypothermia. Up to this point a total of 572 patients received a kidney transplant (285 in the hypothermia group and 287 in the normothermia group). 28% of recipients in the hypothemia group developed delayed graft function compared to 39% in the normothermia group.
The authors therefore conclude that mild hypothermia significantly reduces the rate of delayed graft functions among recipients.
- This study suggests that potential organ donors after declaration of death according to neurologic criteria should be treated with mild hypothermia.
- Intensive care units that continue to treat patients with mild hypothermia after cardiac arrest might have two rewarm their patients for the diagnosis of neurological death before re-cooling them for organ transplantation
Anyhow, it seems reasonable not to get rid of your cooling devices!

The Targeted Temperature Management Trial: Nielsen N, et al. New Engl J Med. 2013 Dec;369(23):2197-206
The 2 trials that introduced therapeutic hypothermia into ICU practice:
The Hypothermia After Cardiac Arrest Study Group, Holzer at al. New Engl J Med. 2002 Feb;346(8):549-556
Bernard S.A. et al. New Engl J Med. 2002 Feb;346(8):557-563
Review article on therapeutic hypothermia for non-VF/VT cardiac arrest:
Sandroni S. et al. Crit Care Med; 2013;17:215
Pyrexia and neurological outcome:
Leary M. et al. Resuscitation. 2013 Aug;84(8):1056-61
BIJC post on: The Effect of Pre-Hospital Cooling: Rather Worrying Results

Since then a couple of well conducted, larger trials have failed to show a positive impact of IABP especially on mortality. In regards of the most recent meta-analysis in JAMA we provide a short overview of the most important publications. It's interesting to see that the balloons undermining started with a meta-analysis and for the the time being ends with one.
Stitch no.1
The first notable hole in the ballon was caused by Sjauw et al.'s systematic review and meta-analysis in the European Heart Journal in 2009. Their pooled randomized data consisting of two separate meta-analyses did not support the use of an IABP in patients with high risk STEMI. They concluded that there is insufficient evidence endorsing the current guideline recommendation for the use of IABP therapy in the setting of STEMI complicated by cardiogenic shock.
This publication was one of the main reasons for the expert panel of the European Society of Cardiology to change the recommendation (ESC Guidelines 2012) to use an IABP in patients with STEMI from IC to IIB.
Stitch 2 and 3
In the same year 2012 Thiele et al. published their first IABP-SHOCK II results in the NEJM. Their
randomized, prospective, open-label, multicenter trial showed no reduction in the 30-day mortality compared to the best available medical therapy alone in patients with myocardial infarction-induced cardiogenic shock and planned early revascularization (PCI or CABG).
One year later the IABP-SHOCK II investigators published their final 12-months results in The Lancet. They came to the final conclusion that in patients undergoing early revascularization for myocardial infarction with cardiogenic shock, IABP did not reduce 12-month all-cause mortality.
Stitch no. 4
In 2013 Ranucci at al. presented the results of their single-center prospective randomized controlled trial looking at the usage of a preoperative IABP in high-risk patients undergoing surgical coronary revascularization. By looking at a total of 110 patients with an ejection fraction below 35% and no hemodynamic instability there was no improvement in outcome when inserting an IABP preoperatively.
Preliminary Final Stitch
So finally Ahmad and his team decided to assess IABP efficacy in acute myocardial infarction by performing an updated meta-analysis. Main outcome was 30-day mortality. They included 12 eligible RCTs randomizing 2123 patients and found no improvement in mortality among patients with acute myocardial infarction... regardless of whether patients had cardiogenic shock or not!
A look at another 15 eligible observational studies with a total of 15 530 patients showed basically conflicting results which was explained by the differences between studies in the balance of risk factors between IABP and non-IABP groups.
It seems that the IABP fails to show its assumed efficacy in patients with myocardial infarction and cardiogenic shock, especially when early revascularization (PCI or CABG) is available.
As a general consideration and also when no early revascularisation is available the use of another left-ventricular assist device like the Impella pump might prove to be a good and easy to use alternative (see blow).
Sjauw KD et al. Eur Heart J 30: 459-468
ESC Guidelines for the management of acute myocardial infarction in patients presenting with ST-segment elevation. Eur Heart J 33: 2569-2619 OPEN ACCESS
Thiele et al. N Engl J Med 2012; 367:1287-1296 OPEN ACCESS
Thiele et al. The Lancet, Volume 382, Issue 9905, Pages 1638 – 1645
Crit Care Med. 2013 Nov;41(11):2476-83
JAMA Intern Med. Published online March 30, 2015
Short film on the principle of the Impella pump 2.5. Bare in mind that this device can actually be easily inserted in the environment of ICU and positioned by using transthoracic echo TTE.

Jefferson et al. used the data of 46 trials with oseltamivir or zanamivir for this review. They basically conclude that:
- Both drugs shorten the duration of symptoms of influenza-like symptoms by less than a day
- Oseltamivir did not affect the number of hospitalizations
- Prophylaxis trials showed a reduced risk of symptomatic influenza in individuals and households, but no definite conclusion can be made
- Oseltamivir use was associated though with nausea, vomiting, headaches, renal and psychiatric events
...and finally write: 'The influenza virus-specific mechanism of action proposed by the producers does not fit the clinical evidence'. This review certainly undermined the importance of oseltamivir for many of us.
The Cochrane review though did not look at outcomes like mortality, but the Lancet Respiratory Medicine did! Stella G at al. have now published a large systematic review which included 29'234 patients from 78 studies during the period from 2009 to 2014. Their findings come rather surprisingly:
- Compared with no treatment, neuraminidase inhibitor treatment (irrespective of timing) was associated with a reduction in mortality risk
- Compared with later treatment, early treatment (within 2 days of symptom onset) was associated with a reduction in mortality risk
- The reduction in mortality risk was observed when treatment was started up to 5 days of symptoms onset
There still seem to be some good reasons to use oseltamivir in critically ill patients with suspected or proven influenza... up to 5 days of symptoms onset!
Jefferson T et al. The Cochrane Collaboration, Published Online: 10 APR 2014
The Cochrane Collaboration News Release 10 April 2014
Muthuri, Stella G et al. The Lancet Respiratory Medicine , Volume 2 , Issue 5 , 395 - 404

ARISE and ProCESS had been published before (read here) and both of them showed no difference between EGDT and 'usual care'.
ProMISe included 1251 patients with severe sepsis or septic shock that were admitted to a total 56 hospitals in the UK. Again classical EGDT with measurement of continuous central venous oxygenation was compared to so called 'usual treatment'. It's remarkable to notice that in the 'usual treatment' group about half of the patient didn't get a central line and central venous oxygenation wasn't even measured in the ones who got one. And here's the result:
There was no difference in 90-day mortality and no differences in secondary outcomes. In contrast EGDT actually increased costs.
It has become difficult to ignore these three trials!
Our conclusion: The classical EGDT therapy has ended here and now... but EGDT will keep its central role in the treatment of septic patients!
Early:
- Identify septic patient quickly, start screening for patients if indicated
- Administer antibiotics within the first our of recognition of sepsis
- Start IV-fluid therapy immediately
- Take (blood) cultures as quick as possible, but do not delay antibiotic treatment
Goal Directed:
- Aim for a reasonable mean arterial pressure (e.g. 65mmHg)
- Aim for a sufficient urinary output (0.5ml/h)
- Central venous pressure (CVP) certainly and most probably central venous oxygenation (ScvO2) are not parameters to measure fluid responsiveness
- Lactate remains an issue of debate
Therapy:
- Simple: Whatever the physician feels is best!
ProMISe Trial, Mouncey et al. N Engl J Med. 2015 Mar 17.
BIJC Review on ARISE and ProCESS
Picture displayed taken from the Ice Cream Trilogy by Wright, Pegg and Frost
Doctors Should Position their Name Tag on the Right Side of their Chest... It's Evidence Based!
23/3/2015
 They're All WRONG!!!
They're All WRONG!!! Every day we all do our best to establish the best patient-physician relation possible. The first impression counts and besides introducing ourselves verbally we ware name tags to visually inform about our name and position. But did anyone actually ever ask himself if it makes a difference wether you ware your name tag on the left or right side of your chest?
Well, an orthopaedic surgeon in Switzerland obviously has and conducted a 'blinded' study to answer this specific question. He made one hundred volunteers, blinded to the experimental setup, present for an orthopedic consultation in a standardized manner. The name tag of the physician was randomly positioned on the left chest side and presented to 50 individuals (age 35 years (range 17 to 83)) or the right chest side and then presented to 50 other individuals (35 years (range 16 to 59)). The time of the participant noticing the name tag was documented. Subsequently, the participant was questioned concerning the relevance of a name tag and verbal self-introduction of the physician.
38% of the participants noticed the nametag on the right as opposed to 20% who noticed it if placed on the left upper chest... hey, this turned out to be statistically significant, giving us a p-value of 0.0473!
The author concludes: Positioning the name tag on the right chest side results in better and faster visibility.
We conclude:
- Also orthopaedic surgeons seem to be interested in a close patient-physician relationship
- Orthopedic surgeons actually do talk to their patients!
- Orthopedic surgeons in Switzerland don't seem to be overstrained with their workload
Schmid SL et al. March 2015, PLOS One, DOI: 10.1371/journal.pone.0119042

- Currently no indications exist for the routine use of colloids over crystalloids
- In regards of current evidence (including the Albios trial), the cost and limited shelf time the use of albumin as a resuscitation fluid is not recommended
- The use of hydroxy-ethyl-starch (HES) during resuscitation should be avoided
- In light of the lack of evidence, and the theoretical potential for adverse effect, the suggestion is to avoid gelatine or dextran
- The use of 0.9% normal saline is associated with the development of hyperchloremic metabolic acidosis and increased risk of AKI in susceptible patients. Therefore balanced crystalloid solutions should be considered/ preferred
- Current literature supports the use of balanced crystalloid solutions (e.g. Hartmann's solution, Ringer's lactate) whenever possible
This makes things quite simple actually... but of course opinions differ!
Lira and Pinsky, Annals of Intensive Care Dec 2014, 4:38 OPEN ACCESS
Read here: The Albios trial
- Platelets must be stored at room temperature
- Because of the risk of bacterial growth the shelf life of platelet units is only 5 days
- Maintaining a constant pool of platelets for clinical work is extremely difficult and resource-intensive
- Transfusion related risks are notable (e.g. febrile reaction 1/14, allergic reactions 1/50, bacterial sepsis 1/75'000)
When it comes to their usage intensivists often have a different approach than haematologists and guidelines mostly vary from hospital to hospital, from country to country. However, instead of searching all the literature yourself you might consider reading the article by Kaufman RM et al. published just this month in the Annals of Internal Medicine.
A panel of 21 specialists, covering almost all areas of medicine involved in handling platelets, performed a systematic review by looking up publications from 1900 to 2013. From 1024 identified studies, 17 RCTs and 53 observational studies were included in the review. The result of their work are guidelines on the use of platelets including their grade of recommendation. Short:
- Platelets should be transfused prophylactically to reduce the risk for spontaneous bleeding in hospitalized adult patients with therapy-induced hypo proliferative thrombocytopenia < 10 x 109 cells/L (Grade: strong recommendation; moderate-quality evidence)
- Platelets should be transfused prophylactically for patients having elective central venous catheter placement with a platelet count less than 20 × 109 cells/L (Grade: weak recommendation; low-quality evidence)
- Prophylactic platelet transfusion is recommended for patients having elective diagnostic lumbar puncture with a platelet count less than 50 × 109 cells/L (Grade: weak recommendation; very low-quality evidence)
- Prophylactic platelet transfusion is recommended for patients having major elective nonneuraxial surgery with a platelet count less than 50 × 109 cells/L (Grade: weak recommendation; very low-quality evidence)
- Routine prophylactic platelet transfusion for patients who are nonthrombocytopenic and have cardiac surgery with cardiopulmonary bypass (CPB) is NOT recommended. Platelet transfusion for patients having CPB who exhibit perioperative bleeding with thrombocytopenia and/or evidence of platelet dysfunction is NOT recommended (Grade: weak recommendation; very low-quality evidence)
- Recommendations for or against platelet transfusion for patients receiving antiplatelet therapy who have intracranial hemorrhage (traumatic or spontaneous) cannot be made (Grade: uncertain recommendation; very low-quality evidence)
It is remarkable to see that after a century of intense research we are left with some moderate-quality evidence and lots of low quality evidence and therefore weak recommendations. I guess guidelines will continue to vary from doctor to doctor, hospital to hospital... country to country.
Kaufman RM et al. Ann Intern Med, Nov 11, 2014: Open access article
Search
Translate
Select your language above. Beware: Google Translate is often imprecise and might result in incorrect phrases!
Categories
All
Airway
Cardiovascular
Controversies
Endocrinology
Fluids
For A Smile ; )
Guidelines
Infections
Meducation
Neurology
Nutrition
Pharmacology
Procedures
Radiology
Renal
Respiratory
Resuscitation
SARS CoV 2
SARS-CoV-2
Sedation
Sepsis
Transfusion
Archives
January 2021
September 2020
March 2020
February 2020
January 2020
December 2019
November 2019
July 2019
May 2019
March 2019
February 2019
January 2019
December 2018
January 2018
October 2017
August 2017
June 2017
March 2017
February 2017
January 2017
October 2016
July 2016
June 2016
April 2016
February 2016
December 2015
October 2015
September 2015
August 2015
July 2015
June 2015
May 2015
April 2015
March 2015
January 2015
December 2014
November 2014
October 2014
September 2014
August 2014
July 2014
June 2014
May 2014
April 2014
March 2014
February 2014
January 2014
December 2013
November 2013
Author
Timothy Aebi







 RSS Feed
RSS Feed


