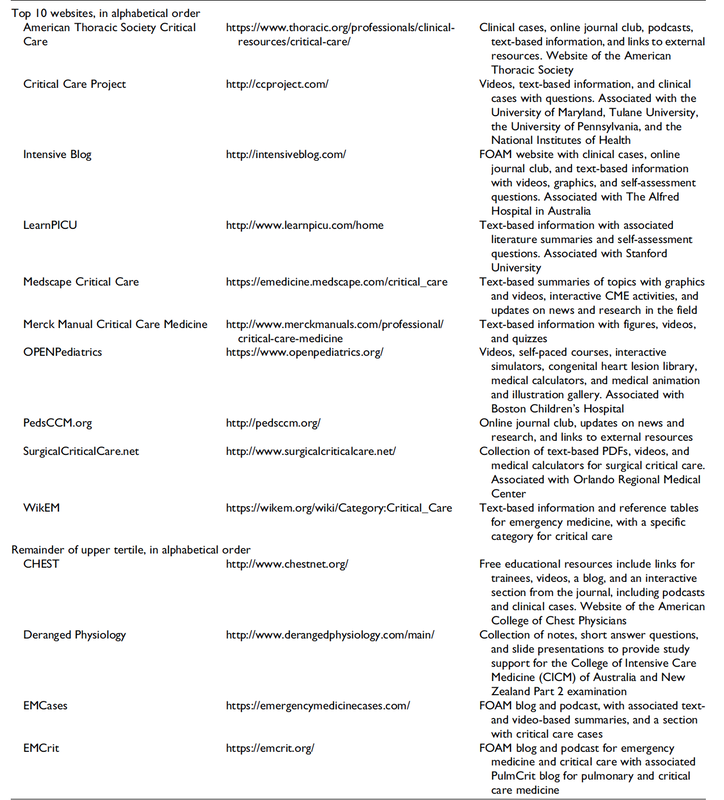
Mind the GAPS Study - Compression Stockings are Useless for Most Elective Surgery Patients!14/9/2020
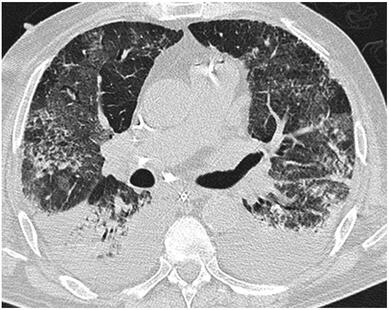
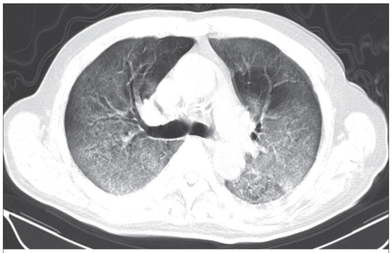
Cricoid pressure prevents aspirations, preoperative antibiotics avoid infections, and compression stockings protect against deep vein thrombosis. Many medical measures aim to reduce morbidity and mortality among patients, but unfortunately, the benefit of these measures is often not, or insufficiently, proven. Under certain circumstances, they may lead to additional problems or even cause harm (e.g. cricoid pressure Read Here). Time has definitely come to take a closer look at compression stockings for surgical patients. Apart from the fact that they look terrible, they are just as uncomfortable to wear and even carry certain risks in patients with peripheral vascular disease, for example. The effectiveness of compression stockings in modern practice has been questioned, but robust evidence has been lacking. This seems to change, as the long-awaited GAPS-Trial has been published and now provides further evidence on what concern patients undergoing elective surgery. Among this population, adding compression stockings to pharmaco-thromboprophylaxis was non-superior compared to pharmaco-thromboprophylaxis alone (primary outcome). There was also no difference in the quality of life outcomes found (secondary outcome). There is now some robust evidence to omit compression stockings in surgical patients that receive pharmacological thromboprophylaxis. Shalhou J. et al. BMJ 2020;369:m1309 The W.H.O. has repeatedly warned that antibiotic resistance is one of the biggest threats to global health today. Among all measures we can take to try and reduce this problem, merely avoiding unnecessary treatments is maybe one of the most effective. It is therefore pleasing that another piece of good evidence has been published, supporting the avoidance of antibiotics in the event of non-complicated diverticulitis (defined as non-perforated diverticulitis with a Hinchey 1a grade in computed tomography). The investigators performed a randomized, placebo-controlled, double-blind trial in which they compared 180 patients with non-complicated diverticulitis to receive either cefuroxime, metronidazole, and amoxicillin/clavulanic acid or placebo. They found No significant difference in the median time of hospital stay (primary outcome). Also, there were no significant differences between groups in adverse events, readmission to the hospital within one week, and readmission to the hospital within 30 days. These findings complement other studies indicating that observational treatment without antibiotics can be considered appropriate in patients with uncomplicated diverticulitis. The lastest updated surviving sepsis guidelines for COVID-19 patient recommends a high-peep strategy in the intubated, mechanically ventilated patient. As most of these patients present with moderate to severe ARDS, PEEP is used to keep lung areas open and therefor to improve oxygenation. This seems to be especially true in the classical case of ARDS, where the lung become 'wet' and 'heavy' which results in widespread atelectasis of the dependent parts of the lungs, often further complicated by pleural effusions. Classical CT appearance in the acute phase of ARDS is an opacification with an antero-posterior density gradient. Dense consolidation in the most dependent regions merges into a background of widespread ground-glass attenuation and the normal or hyperexpanded lung in the non-dependent areas (Howling SJ et al. Clin Radiol 1998;53(2):105-109). The theory behind these changes is that the increased weight of overlying lung causes compression-atelectasis posteriorly. The fact that prone positioning these patients quickly redistributes these gradients supports this theory (Desai SR et al. Anaesthesiology 1991;74(1):15-23). Chest CT's in patients with COVID-19 often show ground-glass opacification with or without consolidations. These are changes often seen in viral pneumonia. Several case series suggest, that CT abnormalities seem to be mostly bilateral and tend to have a peripheral distribution, often involving the lower lobes. In contrast to the classical ARDS pleural thickening, pleural effusion and lymphadenopathy seem to be a less common finding (Shi H et al. Lancet Infect Dis 2020). The leading problem in COVID-19 patients with ARDS is hypoxemia, while hypercapnia does not seem to be a significant problem. Sometimes profound hypoxemia does not seem to correlate with patient symptoms at all. In regards to the images above, atelectasis might not be the predominant reason for V/Q mismatches in these patients. Observations of mechanically ventilated patients in our unit and other hospitals in Switzerland have shown, that higher PEEP levels (15cmH2O and higher) often result in significantly reduced compliance values complicating ventilation and favouring the development of pulmonary over-inflation. This observation might support the theory that patients with COVID do not represent the traditional manner of ARDS with distinctive atelectasis. Another observation that supports this theory is that COVID-19 patients often do not respond as clearly to Prone Positioning as classical ARDS patients do. More probably, V/Q mismatch seems so happen on a more microscopical level in COVID-Patients. Lung compliance is often normal on these patients and, therefore, applying high PEEP-levels does NOT add any benefit at all. Maybe the principle of less is more also applies to COVID-19 patients we treat (Gattinoni L et al. Intensive Care Medicine; 46, pages780–782(2020)) Looking at the New Surviving Sepsis Campain COVID-19 Guidelines: Given these considerations, the strategy with High PEEP-levels in general should be questioned in principle.  The International Liaison Committee on Resuscitation has published the last guidelines for advanced cardiac life support (ACLS) on resuscitation ILCOR in 2015. Usually, these statements are updated every five years, but 'Circulation' has now published an AHA (American Heart Association) focused update due to an increased number of studies looking at ACLS-specific interventions. These updates are focused on three specific areas:
No News in regards to Vasopressors and ECPR Vasopressors in Cardiac Arrest
The bottom line: Great, these recommendations are no real news and do not change current guidelines at all. Extracorporeal Cardiopulmonary Resucitation ECPR
The bottom line: ECPR is not for on the roads and remains an exception in general. Advanced Airway Management Taking recent evidence into account the updated guidelines 2019 conclude:
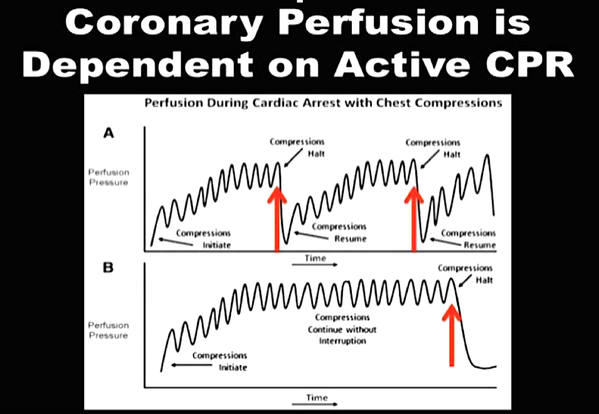
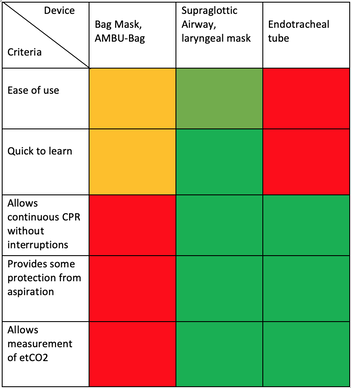
We Suggest: Put the Supraglottic Airway First!In regards to these updated guidelines, the necessity of optimal cardiopulmonary resuscitation (CPR) during resuscitation and practical considerations, it seems reasonable to put the supraglottic airway (SGA) to the very top of airway management! Here is why:
On the other hand
It, therefore, seems plausible to put the supraglottic airway first. Not only first as a choice of airway management, but also one of the first things to do:
The International Liaison Committee on Resuscitation (ILCOR) has again carried together all evidence and recently published more than 50 new ILCOR treatment recommendations and scoping reviews. You can find these documents right here: https://costr.ilcor.org This website provides an excellent systematic review of the Advanced Airway Management during Adult Cardiac Arrest, containing references to all relevant evidence available. Based on this and given the experience from everyday clinical practice, it would be worth considering supplementing the recommendations as follows. - For resuscitation performed by health care professionals (physicians, nurses, paramedics), the use of a supraglottic airway (ideally non-inflatable) as soon as possible is recommended. 2019 AHA Focused Updated on Adult Cardiovascular Life Support The headlines in the news 2017 were remarkable indeed: "Doctor believes he has found the cure for sepsis..." or "Doctor says improvised 'cure' for sepsis has had remarkable results". Dr. Paul Marik described his observation in an interview in 2017, where he mentions several cases of sepsis that have almost miraculously responded to the application of vitamin c (watch here: Interview on WAVY TV). He even continues, that since then they see "the same thing over and over again". This implicated that these results were reproducible. He finally stated that the current data at that stage were "impressive" and that there was enough basic science to show that it works. Vitamin C has many interesting properties that theoretically could be on benefit in sepsis. (read here: Crit☁ post on Vitamin C). Its application was already proposed for the treatment of other diseases like the common cold of Influenza. Despite some moderate positive influence observed, these results could not be reproduced in trials. While the news picked up on this story as a miracle drug, Paul Marik et al. published their results of a before-and-after single-centre, retrospective cohort study in Chest 2017. In this paper, they compared 47 patients with sepsis that received the metabolic cocktail (Vitamin C 1.5g 6-hourly, hydrocortisone 50mg 6-hourly and thiamine 200mg 12-hourly) to 47 patients which did not - notably in a non-double-blinded, non-randomized fashion. Their results showed overall hospital mortality of 8.5% with the 'cocktail' and 40.4% without its application. This publication was reason enough to launch a small war of faith about sense and nonsense of this cocktail for sepsis. The VITAMINS Trial - First FailureSince 2017 a tiny bunch of studies were published, many of them with significant limitations like a small number of patients, often not randomized-controlled and with conflicting results. Nabil Habib T, Ahmed I (2017) Early Adjuvant Intravenous Vitamin C Treatment in Septic Shock may Resolve the Vasopressor Dependence. Int J Microbiol Adv Immunol. 05(1), 77-81. Shin et al. J Clin Med. 2019 Jan; 8(1): 102. Fowler et al. JAMA. 2019 Oct 1;322(13):1261-1270. Fujii et al. have just now published the first more substantial and rigorous trial taking a closer look at the influence on vitamin c in sepsis. They performed an international, multicenter, randomized-controlled open label trial In which they enrolled 211 patients with septic shock admitted to an ICU. They compared Treatment with Vitamin C 1.5g 6-hourly IV, hydrocortisone 50mg 6-hourly IV and thiamin 200mg 12-hourly IV to Hydrocortisone 50mg 6-hourly IV only. They found No difference in time alive and time free of vasopressors (primary endpoint) and No difference 28 days or 90 days mortality (secondary endpoint) This first study on a larger scale, unfortunately, disappoints. More trials are on the way and might give a clearer picture of this topic to come to a final decision eventually. For the moment it is appropriate to state:
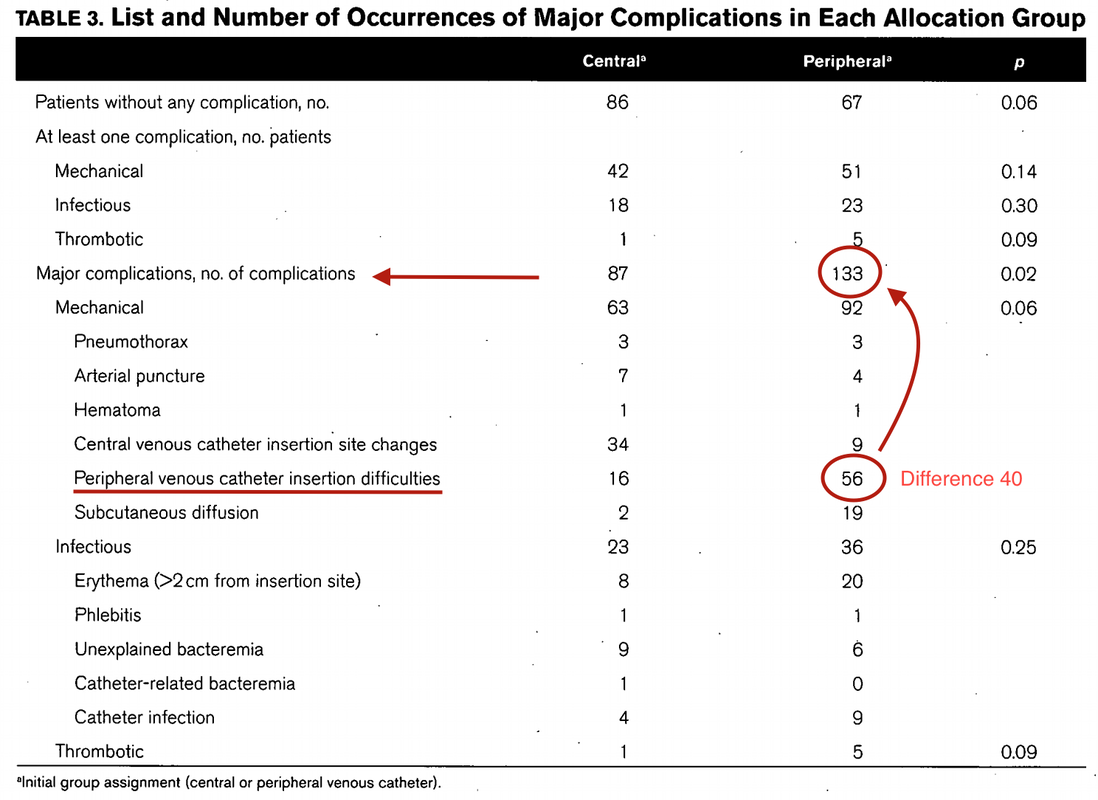
Just as a reminder: Guidelines recommend against the routine use of glucocorticoids in patients with sepsis. However, corticosteroid therapy is appropriate in patients with septic shock that is refractory to adequate fluid resuscitation and vasopressor administration. Fujii et al. JAMA. Published online January 17, 2020. doi:10.1001/jama.2019.22176 Teaching in medical school and opinions in literature are in agreement: The application of vasopressors requires central venous access. The reason for this are concerns that vasopressors given over a peripheral venous catheter (PCV) may cause phlebitis or even worse necrosis or ischemia through extravasation. While irritation of a peripheral vein is often observed with the administration of drugs like potassium or amiodarone, this usually is not the case with the application of, e.g. norepinephrine. Besides, it is essential to keep in mind that the insertion of a central venous catheter (CVC) is technically demanding and takes a certain amount of time when performed correctly. The procedure is also associated with potentially dangerous complications that might be hazardous to the patient. Therefore a fundamental question arises: Do all patients that require vasopressors need a central venous catheter? What about the peripheral access (PVC) - Any dangers there?
|
Search
|






























 RSS Feed
RSS Feed


