 When performing a kidney transplantation nowadays up to 50% of recipients developed a delayed graft function which is defined as the need of dialysis within seven days. The authors of this recently published NEJM-article asked themselves whether mild hypothermia might influence outcome in this regard. In order to answer this question the investigators assigned organ donors after declaration of death according to neurologic criteria into two groups. They were either treated with mild hypothermia (34 to 35°C) or with normothermia (36.5 to 37.5°C). The target temperature was maintained until the patients were transferred to theatre for transplantation. Primary outcome of this trial was delayed graft function among recipients. Secondary outcomes included the rates of individual organs transplanted into each treatment group at the total number of organs transplanted from each donor. This trial had to be stopped early as an interim analysis showed significant efficacy of mild hypothermia. Up to this point a total of 572 patients received a kidney transplant (285 in the hypothermia group and 287 in the normothermia group). 28% of recipients in the hypothemia group developed delayed graft function compared to 39% in the normothermia group. The authors therefore conclude that mild hypothermia significantly reduces the rate of delayed graft functions among recipients.
Anyhow, it seems reasonable not to get rid of your cooling devices!  Read more about the controversies of hypothermia in the ICU: The Targeted Temperature Management Trial: Nielsen N, et al. New Engl J Med. 2013 Dec;369(23):2197-206 The 2 trials that introduced therapeutic hypothermia into ICU practice: The Hypothermia After Cardiac Arrest Study Group, Holzer at al. New Engl J Med. 2002 Feb;346(8):549-556 Bernard S.A. et al. New Engl J Med. 2002 Feb;346(8):557-563 Review article on therapeutic hypothermia for non-VF/VT cardiac arrest: Sandroni S. et al. Crit Care Med; 2013;17:215 Pyrexia and neurological outcome: Leary M. et al. Resuscitation. 2013 Aug;84(8):1056-61 BIJC post on: The Effect of Pre-Hospital Cooling: Rather Worrying Results  Beverley Hunt at al. have just published an excellent practical guideline for the haematological management of major haemorrhage which also serves a a great educational review on this topic... an excellent piece of work! The authors look at this topic point for point and review current literature in an easy to understand sort of manor. They define major blood loss when it leads to a heart rte of >110/Min or a systolic blood pressure of less than 90mmHg, or simply said: when bleeding becomes haemodynamic relevant. In general it is recommended to have a major haemorrhage protocol at hand (1D) and all staff should be trained to recognise major blood loss early (1D). Here's a summary of the recommendations made by the British Committee for Standards in Haematology (BCSH): In Major Haemorrhage.... Red Blood Cells RBC - Hospitals must be prepared to provide emergency Group 0 red cells and group specific red cells (1C) - Patients must have correctly labelled samples taken before administration of emergency Group 0 blood (1C) - There is NO indication to request 'fresh' or 'young' red cells (under 7d of storage, 2B) - Note: The optimum target haemoglobin concentration (Hb) in this clinical setting in general is NOT established. Current literature shows a tendency towards restriction towards 70-90g/L, but the BCSH makes no recommendations therefore (see blow) Cell Salvage (e.g. cell saver) - 24h access to cell salvage should be available in cardiac, obstetric, trauma and vascular centres (2b) Haemostatic Monitoring - Use haemostatic tests regularly during haemorrhage, every 30-60min, depending on severity of blood loss (1C) - Measure platelet count, PT, aPTT (1C) - Note: The BCSG does not recommend TEG and ROTEM at this stage Fresh Frozen Plasma FFP - Use FFP in a 1:2 ratio with RBC initially (2C) - Once bleeding is under control administer FFP when PT and/or aPTT is >1.5 times normal (recommended dose 15-20ml/kg, 2C) - The use of FFP should not delay fibrinogen supplementation if necessary (2C) Fibrinogen - Supplement fibrinogen when levels fall below 1.5g/L Prothrombin Complex Concentrates PCC - Do not use PCC Platelets - Keep the platelet count >50 x 10^9/L (1B) - If bleeding persists give platelets if count falls below 100 x 10^9/L (2C) Tranexamic Acid TA - Give tranexamic acid as soon as possible to patients with, or at risk of major haemorrhage (Recommended dose: 1g IV over 10min, followed by 1g IV over 8h, 1A) - Note: TA has no known adverse effects - Note: Aprotinin is not recommended Recombinant Activated Factor VIIa (Novo Seven) - Do not use Specific Clinical Situations Obstetrics - Fibrinogen levels increase during pregnancy to 4-6g/L - In major obstetric haemorrhage fibrinogen should be given when levels are <2.0g/L (1B) GI-Bleed - Use restrictive strategy for RBC transfusion is recommended in most patients (1A) Trauma - Transfuse adult trauma patients empirically with a 1:1 ratio of FFP : RBC (1B) - Consider early use of platelets (1B) - Give tranexamic acid as soon as possible (Dose 1g over 10min and then 1g over 8h, 1A) Prevention of Bleeding in High-Risk Surgery - Use tranexamic acid (Dose 1g over 10min and then 1g over 8h, 1B) Hunt B et al. British J Haemat, July 6 2015 Read more HERE: Great Review on Transfusion, Thrombosis and Bleeding Management Restricitve Transfusion Threshold in Sepsis, the TRISS Trial Transfusion: Harmful for Patients Undergoing PCI?  A number of things we do for patients in the ICU we simply do... because it has been always done. Maybe because it seems to make sense or because we were simply taught to do so. One of these treatments is the application of oxygen to patients suffering of myocardial infarction. But in fact the impact of this measure is not that well established and we know that high concentrations of oxygen can actually be harmful. Some previous studies suggest possible increase in myocardial injury. Stub et al. therefore performed a multicenter, prospective, randomised controlled trial comparing oxygen (8 L/min) with no supplemental oxygen in patients with STEMI diagnosed on paramedic 12-lead electrocardiogram. They finally included 441 patients into their study. Their primary endpoint was evaluation of infarction size assessed by the cardiac enzymes troponin (cTnI) and creatine kinase (CK). While troponin levels did not differ there was a significant difference in the mean peak CK levels, being higher in the group with oxygen applied. By looking at the secondary endpoints they also found an increase in the rate of recurrent myocardial infarctions and in the frequency of cardiac arrhythmia among the oxygen group. Finally at 6-months the oxygen group had an increase in myocardial infarct size on cardiac MRI. They conclude that patients with myocardial infarction but without hypoxemia may actually not benefit of supplemental oxygen therapy. Time to question our automatisms when treating patients. Stub et al. Circulation. 2015 Jun 16;131(24):2143-50.  In 2013 a small open label, randomised, controlled trial in the NEJM hit the headlines: Nood et al. were able to show that the enteral infusion of donor feces is a potential therapeutic strategy against recurrent C. difficile infections. This was important news as the efficacy of antibiotic therapy decreases with subsequent recurrences. The study suggested that the enteral infusion of donor feces, as compared with vancomycin therapy, results in much better treatment outcomes. But as we all know, every treatment has its side effects. Nood et al. described diarrhoea, cramping and belching as observed adverse events, but all of this resolved quite quickly... Alang et al. have now published a brief report of a woman with recurrent C. difficile infections despite treatments with Metronidazol and Vancomycin. Despite all efforts diarrhoea re-occured again and she finally underwent fecal microbiota transplantation. After the application of donor stool form her daughter through the colonoscope her symptoms resolved and no further C. difficile infection occurred. 16 months later the patient presented again with an unintentional weight gain of about 15kg. The authors mention that several factor might have contributed to her weight gain (e.g. concurrent treatment of existing H. pylori). In regards of the fact that the patient had never been obese before and the donor also experienced a significant weight gain raises the possibility that the fecal transplantation might have at least partially caused her obesity. And important to know: Animal models have shown, that an obese microbiota can be transmitted. Looks like Newport Hospital in Rhode Island took the appropriate action: their policy now is to use non-obese donors only. Along et al. Open Forum Infectious Diseases, Nov 2014 OPEN ACCESS Nood et al. N Engl J Med 2013;368:407-15. OPEN ACCESS  The European Society for Clinical Microbiology and Infectious Diseases has now released new guidelines on the diagnosis and treatment of biofilm infections. Written for clinical microbiologists and infectious disease specialists this paper is a MUST READ for anyone involved in treating critically ill patients. These guidelines outline the nature and properties of biofilms and and their implications on mostly chronic infections caused. As biofilms are very common in critically ill patients it is important to know what specific problems you might encounter, how to proceed and perform a proper diagnosis and what are the essential bits and pieces in the prevention and treatment of biofilm infections. The article is OPEN ACCESS: Clin Microbiol Infect. 2015 Jan 14. pii: S1198-743X(14)00090-1.  In the late 1960's the technology of counter-pulsation by using an intra-aortic balloon pump (IABP) was introduced into clinical work. Based on the principle of diastolic inflation and systolic deflation, IABP counter-pulsation improves diastolic coronary artery blood flow and decreases left ventricular afterload. Up to the year 2009, 2012 respectively, the usage of an IABP in patients with ST-segment elevation myocardial infarction and cardiogenic shock was considered a class IC recommendation (reminder: levels of evidence). Since then a couple of well conducted, larger trials have failed to show a positive impact of IABP especially on mortality. In regards of the most recent meta-analysis in JAMA we provide a short overview of the most important publications. It's interesting to see that the balloons undermining started with a meta-analysis and for the the time being ends with one. Stitch no.1 The first notable hole in the ballon was caused by Sjauw et al.'s systematic review and meta-analysis in the European Heart Journal in 2009. Their pooled randomized data consisting of two separate meta-analyses did not support the use of an IABP in patients with high risk STEMI. They concluded that there is insufficient evidence endorsing the current guideline recommendation for the use of IABP therapy in the setting of STEMI complicated by cardiogenic shock. This publication was one of the main reasons for the expert panel of the European Society of Cardiology to change the recommendation (ESC Guidelines 2012) to use an IABP in patients with STEMI from IC to IIB. Stitch 2 and 3 In the same year 2012 Thiele et al. published their first IABP-SHOCK II results in the NEJM. Their randomized, prospective, open-label, multicenter trial showed no reduction in the 30-day mortality compared to the best available medical therapy alone in patients with myocardial infarction-induced cardiogenic shock and planned early revascularization (PCI or CABG). One year later the IABP-SHOCK II investigators published their final 12-months results in The Lancet. They came to the final conclusion that in patients undergoing early revascularization for myocardial infarction with cardiogenic shock, IABP did not reduce 12-month all-cause mortality. Stitch no. 4 In 2013 Ranucci at al. presented the results of their single-center prospective randomized controlled trial looking at the usage of a preoperative IABP in high-risk patients undergoing surgical coronary revascularization. By looking at a total of 110 patients with an ejection fraction below 35% and no hemodynamic instability there was no improvement in outcome when inserting an IABP preoperatively. Preliminary Final Stitch So finally Ahmad and his team decided to assess IABP efficacy in acute myocardial infarction by performing an updated meta-analysis. Main outcome was 30-day mortality. They included 12 eligible RCTs randomizing 2123 patients and found no improvement in mortality among patients with acute myocardial infarction... regardless of whether patients had cardiogenic shock or not! A look at another 15 eligible observational studies with a total of 15 530 patients showed basically conflicting results which was explained by the differences between studies in the balance of risk factors between IABP and non-IABP groups. It seems that the IABP fails to show its assumed efficacy in patients with myocardial infarction and cardiogenic shock, especially when early revascularization (PCI or CABG) is available. As a general consideration and also when no early revascularisation is available the use of another left-ventricular assist device like the Impella pump might prove to be a good and easy to use alternative (see blow). Sjauw KD et al. Eur Heart J 30: 459-468 ESC Guidelines for the management of acute myocardial infarction in patients presenting with ST-segment elevation. Eur Heart J 33: 2569-2619 OPEN ACCESS Thiele et al. N Engl J Med 2012; 367:1287-1296 OPEN ACCESS Thiele et al. The Lancet, Volume 382, Issue 9905, Pages 1638 – 1645 Crit Care Med. 2013 Nov;41(11):2476-83 JAMA Intern Med. Published online March 30, 2015 Short film on the principle of the Impella pump 2.5. Bare in mind that this device can actually be easily inserted in the environment of ICU and positioned by using transthoracic echo TTE.  Almost exactly one year ago the Cochrane Library published an intervention review on the prevention and treatment of influenza with neuraminidase inhibitors in adults and children. The reason for this review was the fact that many countries stockpile these drugs and the WHO classified them as an essential medicine. Jefferson et al. used the data of 46 trials with oseltamivir or zanamivir for this review. They basically conclude that: - Both drugs shorten the duration of symptoms of influenza-like symptoms by less than a day - Oseltamivir did not affect the number of hospitalizations - Prophylaxis trials showed a reduced risk of symptomatic influenza in individuals and households, but no definite conclusion can be made - Oseltamivir use was associated though with nausea, vomiting, headaches, renal and psychiatric events ...and finally write: 'The influenza virus-specific mechanism of action proposed by the producers does not fit the clinical evidence'. This review certainly undermined the importance of oseltamivir for many of us. The Cochrane review though did not look at outcomes like mortality, but the Lancet Respiratory Medicine did! Stella G at al. have now published a large systematic review which included 29'234 patients from 78 studies during the period from 2009 to 2014. Their findings come rather surprisingly: - Compared with no treatment, neuraminidase inhibitor treatment (irrespective of timing) was associated with a reduction in mortality risk - Compared with later treatment, early treatment (within 2 days of symptom onset) was associated with a reduction in mortality risk - The reduction in mortality risk was observed when treatment was started up to 5 days of symptoms onset There still seem to be some good reasons to use oseltamivir in critically ill patients with suspected or proven influenza... up to 5 days of symptoms onset! Jefferson T et al. The Cochrane Collaboration, Published Online: 10 APR 2014 The Cochrane Collaboration News Release 10 April 2014 Muthuri, Stella G et al. The Lancet Respiratory Medicine , Volume 2 , Issue 5 , 395 - 404  The mainstay of asthma treatment is the beta-2-mimetic salbutamol (or albuterol) which is also first line in critical care. Researchers have previously observed that inhaled glucocorticosteroids potentiated salbutamol-induced airway vascular smooth muscle relaxation. As a consequence of these observations Eliana S. et al. performed a small double-blind pilot study on fifteen patients clinical obstructive asthma. The influence of inhaled steroids (mometasone) compared to placebo 30min before inhalation of salbutamol was measured by looking at the change in FEV1 (ΔFEV1) for airway smooth muscle and the airway blood flow (ΔQaw) for airway vascular smooth muscle within 15 min after drug inhalation. This pilot study showed that in adult patients with obstructive asthma with airflow obstruction, a single standard dose of an inhaled steroid can acutely increase the FEV1 response to a standard dose of inhaled salbutamol... especially when administered 30min before. This might become a helpful finding in the future! We'll keep the eyes open! Eliana S. et al. Chest. 2015;147(4):1037-1042.  Dexmedetomidine has become increasingly popular in critical care and is an attractive alternative to standard sedation drugs like midazolam and propofol. Especially in the context of moderate to light sedation and when weaning the patient form mechanical ventilation. There are two European randomized double-blind studies (PRODEX, MIDEX) showing that dexmedetomidine is non inferior to propofol and midazolam in maintaining target sedation levels in mechanically ventilated intensive care unit patients. Additionally, dexmedetomidine shortened the time to extubation versus both standard sedatives, suggesting that it may reduce ICU resource needs and thus lower ICU costs. The authors therefore decided to take a closer look at the cost factor by performing a secondary, cost-minimization analysis assessing the economics of dexmedetomidine versus standard care sedation. Without going into details it seems that dexmedetomidine actually reduces costs in intubated patients with light to moderate sedation... and this mainly by reducing the time to extubation. It is noteworthy that all these data and their conclusions derive from the same international research team and that the pharmaceutical company providing dexmedetomidine was involved by sponsoring. Nevertheless, these robust results indicate: - Dexmedetomidine is an attractive sedative for moderate to light sedation in the intubated patient - Seems to shorten time to extubation - ... and might actually be cheaper compared to 'standard' sedation Turinen H et al. Critical Care 2015, 19:67 OPEN ACCESS Jacob SM et al. JAMA. 2012;307(11):1151-1160. OPEN ACCESS  As every child already knows by now the study by Rivers et al. in 2002 has raised the awareness about sepsis and led to the establishment of the surviving sepsis campaign. As we have posted on BIJC before, many elements of the early goal directed therapy (EGDT) have been discussed controversially since. In order to answer some of the questions of sepsis treatment three big trials have been started, involving different parts of this world. These efforts have led to a unique situation as we now have three high quality trials looking at the classical EGDT versus 'usual care'. ARISE and ProCESS had been published before (read here) and both of them showed no difference between EGDT and 'usual care'. ProMISe included 1251 patients with severe sepsis or septic shock that were admitted to a total 56 hospitals in the UK. Again classical EGDT with measurement of continuous central venous oxygenation was compared to so called 'usual treatment'. It's remarkable to notice that in the 'usual treatment' group about half of the patient didn't get a central line and central venous oxygenation wasn't even measured in the ones who got one. And here's the result: There was no difference in 90-day mortality and no differences in secondary outcomes. In contrast EGDT actually increased costs. It has become difficult to ignore these three trials! Our conclusion: The classical EGDT therapy has ended here and now... but EGDT will keep its central role in the treatment of septic patients! Early: - Identify septic patient quickly, start screening for patients if indicated - Administer antibiotics within the first our of recognition of sepsis - Start IV-fluid therapy immediately - Take (blood) cultures as quick as possible, but do not delay antibiotic treatment Goal Directed: - Aim for a reasonable mean arterial pressure (e.g. 65mmHg) - Aim for a sufficient urinary output (0.5ml/h) - Central venous pressure (CVP) certainly and most probably central venous oxygenation (ScvO2) are not parameters to measure fluid responsiveness - Lactate remains an issue of debate Therapy: - Simple: Whatever the physician feels is best! ProMISe Trial, Mouncey et al. N Engl J Med. 2015 Mar 17. BIJC Review on ARISE and ProCESS Picture displayed taken from the Ice Cream Trilogy by Wright, Pegg and Frost Doctors Should Position their Name Tag on the Right Side of their Chest... It's Evidence Based!23/3/2015
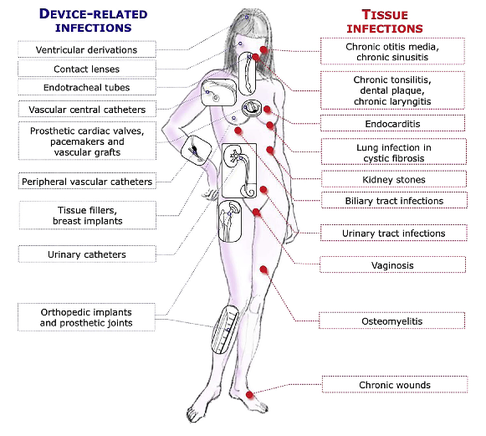
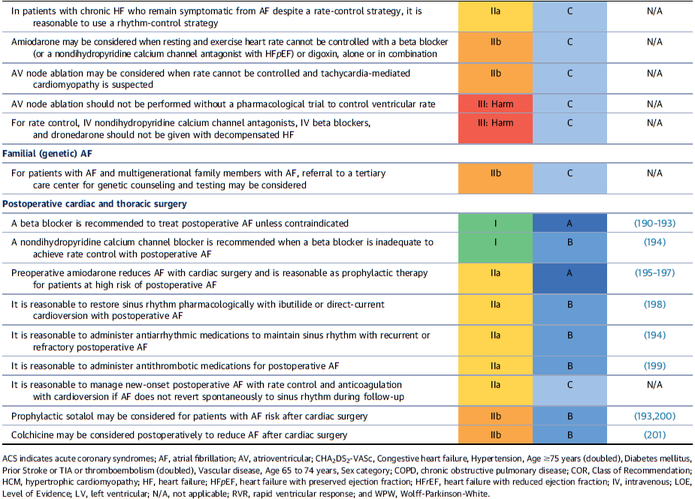
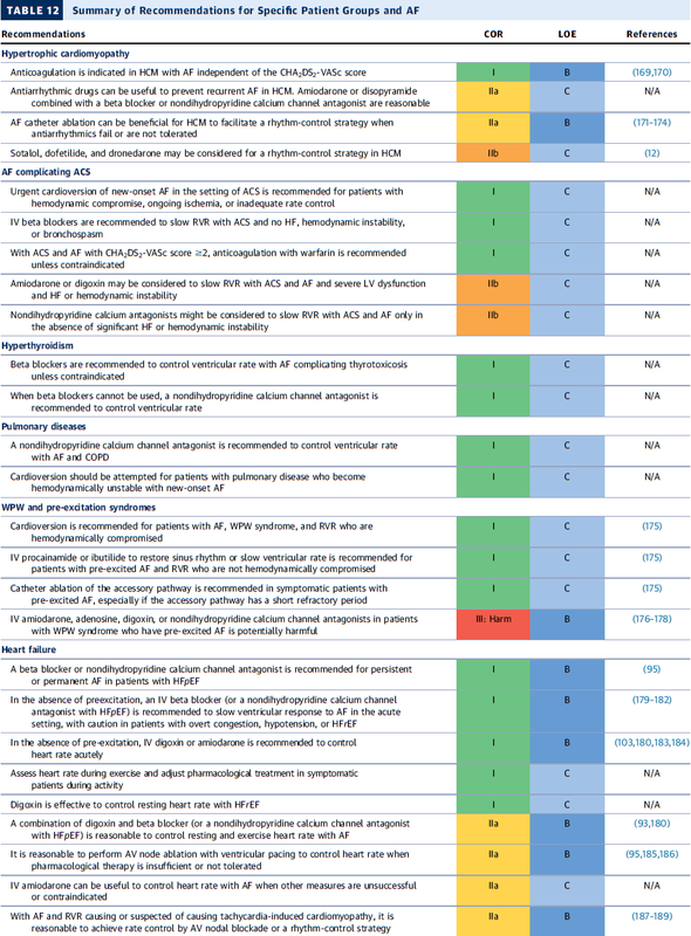
 They're All WRONG!!! They're All WRONG!!! Here it is finally, the study we all have been waiting for. 'THE question' that has never been answered... until now! Every day we all do our best to establish the best patient-physician relation possible. The first impression counts and besides introducing ourselves verbally we ware name tags to visually inform about our name and position. But did anyone actually ever ask himself if it makes a difference wether you ware your name tag on the left or right side of your chest? Well, an orthopaedic surgeon in Switzerland obviously has and conducted a 'blinded' study to answer this specific question. He made one hundred volunteers, blinded to the experimental setup, present for an orthopedic consultation in a standardized manner. The name tag of the physician was randomly positioned on the left chest side and presented to 50 individuals (age 35 years (range 17 to 83)) or the right chest side and then presented to 50 other individuals (35 years (range 16 to 59)). The time of the participant noticing the name tag was documented. Subsequently, the participant was questioned concerning the relevance of a name tag and verbal self-introduction of the physician. 38% of the participants noticed the nametag on the right as opposed to 20% who noticed it if placed on the left upper chest... hey, this turned out to be statistically significant, giving us a p-value of 0.0473! The author concludes: Positioning the name tag on the right chest side results in better and faster visibility. We conclude: - Also orthopaedic surgeons seem to be interested in a close patient-physician relationship - Orthopedic surgeons actually do talk to their patients! - Orthopedic surgeons in Switzerland don't seem to be overstrained with their workload Schmid SL et al. March 2015, PLOS One, DOI: 10.1371/journal.pone.0119042  In December 2014 the AHA, ACC and HRA have released a new bunch of guidelines on the management of patients with atrial fibrillation (AFib). The paper itself is worth reading as it looks into the basic understanding of this condition, its clinical evaluation and finally the treatment options. As these guidelines are open access it can be considered mandatory Free Open Access Meducation FOAMed. Below is a summary of the Recommendations according to specific patient groups. It's interesting to notice that digoxin still plays a role in patients with heart failure, especially when looking at the findings of Turakhia et al. in JACC, Aug 19 2014. J Am Coll Cardiol. 2014;64(21):2246-2280 OPEN ACCESS BIJC post on dixogin in critical care 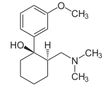 Developed in the early 70'ies tramadol has become a very popular drug for pain relief for various reasons. Among others it is often said that tramadol is safe to use and has non-addictive properties, making this an ideal opioid to use for in and out of hospital. The facts though point in the opposite direction. In JAMA Internal Medicine Fournier et al. have just published a case control analysis to look at the fact that tramadol before has been associated with the occurrence of significant hypoglycemia. Their cohort included a total of 334'034 patients whereas each case of hospitalization for hypoglycemia was matched with up to 10 controls on age, sex, and duration of follow-up. Basically they compared similar patients which were either started on tramadol or codeine for pain treatment. They were able to show that compared with codeine, tramadol use was associated with an increased risk of hospitalization for hypoglycemia, particularly in the first 30 days of use. It has to be noted though, that the overall incidence is low with 7 per 10'000 per annum. In the same issue's commentary Nelson and Juurlink take the opportunity to point out some other remarkable problems associated with Tramadol, again showing us that things are not a simple as we think they are. - Tramadol itself has only a low affinity to opioid receptors and mainly works over one of its metabolites: O-Desmethyltramadol (M1), which then binds to µ opioid receptors - The expression of the enzyme that metabolites tramadol to M1 is extremely variable, thus: giving a certain dose of tramadol leaves you with an unknown dose of acting opioid! - Despite suggestions to the contrary, tramadol does pose a risk for addiction - And there are increasing reports of deaths involving this drug - Other documented adverse effects are: serotonin syndrome and seizures Conclusion: Tramadol remains a non-ideal drug in the setting of an ICU. Fournier et al. JAMA Intern Med. 2015;175(2):186-193. Nelson and Juurlink JAMA Intern Med. 2015;175(2):194-195.  Microbiologically confirmed ventilator-associated pneumonia (VAP) or ventilator-associated conditions (VAC, e.g. worsening oxygenation) in intubated patients remains a major concern in ICU's. VAP is defined as a hospital-acquired pneumonia which develops within 48-72 hours after endotracheal intubation. To prevent this complication ICU's uniformly have adapted the VAP-bundle, a bunch of measures aiming to prevent ventilator-associated pneumonia. Unfortunately the evidence of the VAP-bundle is not as robust as one might think it is. Here's the evidence of some elements of the VAP bundle: - Elevation of the head to bed 45° (low evidence) - Daily sedation interruptions (the impact on reducing VAP has not been shown so far) - Daily oral chlorhexidine rinses (low evidence) ... it's most likely the combination of measures that is of benefit to the patient... hopefully! But hold on, there is another intervention that finally brings quite some evidence with it! Active suctioning of the subglottic area, where nasal-oral secretions gather and create a rich culture medium for all sorts of micro-organisms, also aims to reduce the incidence of VAP. In contrast to the classical VAP-bundle the evidence here is strongly in favour for these devices! In 2005 four registrars in cardiothoracic surgery looked into this topic and summarised their efforts online on Best Evidence Topics, best bets.org. In this blog they review 13 relevant articles on the use of subglottic suctioning and conclude: subglottic suction significantly reduces the incidence of VAP in high risk patients - which means a NNT of 8 if ventilated for more than 3 days. They also mention that this measure is cost effective, despite the more expensive tubes. In the same year Dezfulian et al. presented a systematic meta-analysis of randomized trials in the American Journal of Medicine. They ended up with 5 studies including 869 patients. They also came to the conclusion that subglottic secretion drainage is effective in preventing VAP in patients expected to be ventilated for more than 72 hours. In 2011 Hallais et al. looked into the issue of cost-effectiveness with a cost-benefit analysis. Even when assuming the most pessimistic scenario of VAP incidence and costs the replacement of conventional ventilation with continuous subglottic suctioning would still be cost-effective. In 2011 Muscedere et al. published an 'official' review article in Critical Care Medicine and also ended up with 13 randomised clinical trial, most of them the same 'BestBETs' had already identified 6 years before. It is therefore not surprising to see that they also found a highly significant reduction in VAP. They were also able to demonstrate a reduction in ICU length of stay and duration of mechanical ventilation, although the strength of this association was weakened by heterogeneity of study results. We finally would like to mention the latest randomised controlled trial on this topic which was published in Critical Care Medicine this January 2015. Damas et al. randomly assigned 352 patients to either receive subglottic suctioning or not. Again sublottic suctioning significantly reduced VAP prevalence and therefore also antibiotic use. At least we have identified one area in critical care where an impressive pile of evidence supporting the use of subglottic suctioning in long-term intubated patients is present... and even better: cost-effective analyses also come out in great favour for this measure! Take-home message: Subglottic suctioning does prevent VAP in patients likely to be ventilated more than (48-) 72 hours and should be used in these situations. Review BestBETs 2005 Dezfulian C et al. Am J Med. 2005 Jan;118(1):11-8 Hallais C. et al. Infect Control Hosp Epidemiol. 2011 Feb;32(2):131-5 Muscedere J et al. Crit Care Med. 2011 Vol. 39, No. 8 Damas P et al. Crit Care Med. 2015 Jan;43(1):22-30  How often have you assessed adrenocortical function in critical illness by measuring cortisol levels or performing an ACTH stimulation test? And how often have you been involved in discussions on whether these tests actually should be done? Venkatesh et al. provide us now with an article on this topic, leaving us with that odd sort of feeling... little we know, much we do! These are the ten false beliefs about cortisol in critically ill patients: 1. A relative adrenal insufficiency (RAI) has been proposed to be associated with worse outcome in critically ill patients. In fact, multiple studies have demonstrated that the reverse is true: higher random cortisol values are associated with a greater mortality in septic shock 2. Total plasma cortisol levels fluctuate significantly. Therefore, the interpretation of a single random total cortisol measurement is problematic. 3. Measuring total plasma cortisol by immunoassays will show different results when same sample is analysed by different assays. 4. ACTH stimulation test: There is no consistent relationship between cortisol response and illness severity in critical illness. Even if RAI is suspected, subgroup analysis of randomised controlled trials of steroids in septic shock do not consistently report a benefit of steroids given. 5. A normal ACTH stimulation test does not out-rule a clinically significant adrenal insufficiency. 6. Total plasma cortisol concentrations do not reflect accurately the bioavailable fraction of plasma free cortisol (PFC). 7. Direct measurement of free cortisol is complex and mostly not available. And.. unfortunately, calculating PFC concentrations using the Coolen’s equation is unreliable in critical illness. 8. In critical illness it is difficult, if not even impossible, to assess the tissue adrenal response based on plasma cortisol levels. 9. Elevated plasma cortisol levels are not only due to an increased production but also to a reduction in cortisol clearance occurring in critical illness. 10. Treating critically ill patients with classical doses of hydrocortisone (50-100mg every 8h) may result in grossly supraphysiological levels of cortisol. Got interested on some further background information? Read here: Venkatesh et al. Intensive Care Med, January 2015 |
Search
|

 RSS Feed
RSS Feed


