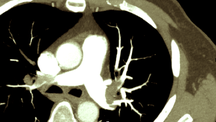
 It's remains a residual concern for clinicians: The fear that intravenous iodinated contrast material used for medical imaging might cause acute kidney injury (AKI), more dialysis and even death. In fact, practice guidelines still tell us to be cautious to use IV contrast material in patients at risk for acute renal failure. In March 2014 a review article in BioMed Research International came to the conclusion, that by reviewing more recent evidence this risk is almost nonexistent in patients with normal renal function. Even in patients with pre-existing renal insufficiency the risk of secondary contrast-induced AKI is probably much smaller than traditionally assumed. Mc Donald et al. have now provided further evidence, that the risk associated with the administration of IV iodinated contrast material has been overstated. In this single-center retrospective study they looked at almost all CT scans from 2000-2010 and, using propensity score matching, created two large groups (approx. 10'000 patients each) to compare patients with enhances CT-scans to the ones without enhanced CT-scans. They were able to show that the rate of emergent dialysis and short-term mortality among the two groups were not significantly different. In fact the development of AKI was independent of contrast material administration. Take home message: - AKI is associated with worse overall short-term outcomes (dialysis, 30-day mortality), but these outcomes are independent of contrast material exposure. The nephrotoxic risk associated with administration of intravenous iodinated contrast material appears to have been overstated. Or short: If you have a good indication for an enhanced CT-scan: Go for it! McDonald RJ et al. Radiology 2014 December OPEN ACCESS Also worth looking at: Davenport MS et al. Radiology 2013 April OPEN ACCESS BIJC post: Iodinated Radiocontrast Agents can cause Kidney Injury... But not as much as we thought they would!  Again we have picked a review article looking at fluid resuscitation in the ICU. This article by Lira et al. in the Annals of Intensive Care looks at all the new literature available in regards of fluid therapy during resuscitation. Also review current recommendations and recent clinical evidence. This results in an excellent systematic review that leaves us with following conclusions: - Currently no indications exist for the routine use of colloids over crystalloids - In regards of current evidence (including the Albios trial), the cost and limited shelf time the use of albumin as a resuscitation fluid is not recommended - The use of hydroxy-ethyl-starch (HES) during resuscitation should be avoided - In light of the lack of evidence, and the theoretical potential for adverse effect, the suggestion is to avoid gelatine or dextran - The use of 0.9% normal saline is associated with the development of hyperchloremic metabolic acidosis and increased risk of AKI in susceptible patients. Therefore balanced crystalloid solutions should be considered/ preferred - Current literature supports the use of balanced crystalloid solutions (e.g. Hartmann's solution, Ringer's lactate) whenever possible This makes things quite simple actually... but of course opinions differ! Lira and Pinsky, Annals of Intensive Care Dec 2014, 4:38 OPEN ACCESS Read here: The Albios trial 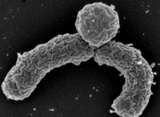 Just recently in 2014 the WHO has requested to develop a draft global action plan to combat emergent antimicrobial resistance (AMR). AMR is present in all parts of the world, new resistance mechanisms emerge and spread globally. And most importantly: Patients with infections by drug-resistant bacteria are generally at risk of worse clinical outcome and death. On the background of this the recent publication in Nature by Ling et al. is remarkable as it might offer the key to a new antimicrobial weapon in the near future. Teixobactin is the name of a macrocylic peptide representing a new class of antibiotics. It appears to be potent bactericidal agent against a broad panel of bacterial pathogens, especially gram-positive bacteria including MRSA, enterococci and VRE as well as M. tuberculosis, C. difficile and Anthrax. Teixobactin inhibits cell wall synthesis and most remarkably showed no development of resistance so far. Teixobactin is produced by E. terrae, a microorganism discovered in the soil of a grassy field in Maine. As mentioned in the article, these 'uncultured' bacteria make up approximately 99% of all species in external environments, and are an untapped source of new antibiotics. An interesting article, especially if you want to see what's going on outside the hospital! Ling LL et al. Nature 2015; doi:10.1038/nature14098  'Anaesthesia', the Journal of the Association of Anaesthetists of Great Britain and Ireland have published an Open Access Supplement on various aspects of Transfusion, Thrombosis and Bleeding Management. This is an excellent opportunity to update your knowledge in this field and actually compulsory for anyone involved actively in critical care. The supplement consists of multiple review articles which are kept nice and short and are perfect for reading in between... In Conclusion: Reading highly recommended! On following website you can find a list of all articles including links to the full text: Anaesthesia, Vol. 70, Issue Supplement s1, January 2015: Transfusion, Thrombosis and Bleeding Management  In 2001 Rivers at al. published their remarkable Study on Early Goal Directed Therapy (EGDT) in Severe Sepsis and Septic Shock. The results of this single centre study were impressive, as this intervention significantly reduced mortality when applied within 6 hours after admission. Despite the implication of these principles into current guidelines many elements of the EGDT are being discussed quite controversially since. Especially goal criteria like central venous pressure (CVP) and mean arterial pressure (MAP) have been increasingly questioned and other elements like the trigger to transfuse red blood cell concentrates and the usage of dobutamine are also on going subjects of discussions. In order to answer some of these questions three big trials were started: - The ProCESS trial (Protocolised Care for Early Septic Shock) Out now! - The ARISE study (Australian Resuscitation in Sepsis Evalutation) Out Now! - The ProMISe study (Protocolised Management of Sepsis, GB) Coming up! ProCESS trial: In this multi-centre study a total of 1531 patients were included and randomised to one of the three following treatment groups: protocol-based EGDT (according to the criteria used in the River's study 2001), protocol based standard treatment (using 6-hours resuscitation instructions with no need for placement of a central venous catheter, administration of inotropes to maintain a certain systolic BP and blood transfusions if haemoglobin level was below 75g/L) or usual care (according to the physician's own assessment). Primary endpoint was 60-day in-hospital mortality. Interestingly the use vasopressors and red blood cell concentrates varied between the groups significantly but there was no difference in mortality by 60 days, 90 days or 1 year. Also the need for organ support showed no difference among the three groups. The ARISe trial is also a multi-centre study where patients with early septic shock were randomly assigned to either receive EGDT or usual care. A total of 1600 patients were included and primary outcome was all-cause mortality within 90 days after randomisation. Also here the two groups showed significant differences in regards of treatment modalities: the EGDT group received larger amounts of IV fluids, were more likely to receive vasopressors, got more re blood cell transfusions and were more likely to be treated with dobutamine. These differences are similar to the ones observed in the ProCESS trial. Again though, there was no difference in all-cause mortality at 90 days. So two high quality trials showed no difference between EGDT and 'usual' care. Does this mean that EGDT is of no use? The answer is: Not at all! Taking into account that Rivers published his study in 2001 many physicians involved in critical care are now 'children of this revolution'. Most intensivists nowadays are trained and aware of the importance of an EARLY treatment. The core message of River's study remains the same: Early treatment is important! It is remarkable though that a protocolled approach is non-superior to the clinical judgement of the treating physician. And I personally feel happy about the fact that protocols don't replace clinical judgement and an individual approach at all. Summarised: The key for a successful sepsis treatment remains the same: Treat EARLY (especially with the administration of antibiotics). And, maybe even more importantly: Simple protocols can be helpful, but DO NOT replace an individual, clinical assessment of the patient with severe sepsis or septic shock. We are looking forward for the results of the third trial pending - ProMISe The Process Investigators, N Engl J Med 2014 The ARISe Investigators, N Engl J Med 2014 Rivers et al. N Engl J Med 2001; 345:1368-1377 Read also: Guidelines Surviving Sepsis Campaign 2008, Dellinger et al. Crit Care Med 2008; 36:296 Guidelines Surviving Sepsis Campaign 2012, Dellinger et al. Crit Care Med 2013; 41:580 BIJC Review: Restricitve Transfusion Threshold is OK in Sepsis - The TRISS Trial  Platelets are used in critical to either prevent or treat bleeding. The problem is that despite all the research and studies we still don't know that much on the best way to clinically use platelets... and they are tricky indeed: - Platelets must be stored at room temperature - Because of the risk of bacterial growth the shelf life of platelet units is only 5 days - Maintaining a constant pool of platelets for clinical work is extremely difficult and resource-intensive - Transfusion related risks are notable (e.g. febrile reaction 1/14, allergic reactions 1/50, bacterial sepsis 1/75'000) When it comes to their usage intensivists often have a different approach than haematologists and guidelines mostly vary from hospital to hospital, from country to country. However, instead of searching all the literature yourself you might consider reading the article by Kaufman RM et al. published just this month in the Annals of Internal Medicine. A panel of 21 specialists, covering almost all areas of medicine involved in handling platelets, performed a systematic review by looking up publications from 1900 to 2013. From 1024 identified studies, 17 RCTs and 53 observational studies were included in the review. The result of their work are guidelines on the use of platelets including their grade of recommendation. Short: - Platelets should be transfused prophylactically to reduce the risk for spontaneous bleeding in hospitalized adult patients with therapy-induced hypo proliferative thrombocytopenia < 10 x 109 cells/L (Grade: strong recommendation; moderate-quality evidence) - Platelets should be transfused prophylactically for patients having elective central venous catheter placement with a platelet count less than 20 × 109 cells/L (Grade: weak recommendation; low-quality evidence) - Prophylactic platelet transfusion is recommended for patients having elective diagnostic lumbar puncture with a platelet count less than 50 × 109 cells/L (Grade: weak recommendation; very low-quality evidence) - Prophylactic platelet transfusion is recommended for patients having major elective nonneuraxial surgery with a platelet count less than 50 × 109 cells/L (Grade: weak recommendation; very low-quality evidence) - Routine prophylactic platelet transfusion for patients who are nonthrombocytopenic and have cardiac surgery with cardiopulmonary bypass (CPB) is NOT recommended. Platelet transfusion for patients having CPB who exhibit perioperative bleeding with thrombocytopenia and/or evidence of platelet dysfunction is NOT recommended (Grade: weak recommendation; very low-quality evidence) - Recommendations for or against platelet transfusion for patients receiving antiplatelet therapy who have intracranial hemorrhage (traumatic or spontaneous) cannot be made (Grade: uncertain recommendation; very low-quality evidence) It is remarkable to see that after a century of intense research we are left with some moderate-quality evidence and lots of low quality evidence and therefore weak recommendations. I guess guidelines will continue to vary from doctor to doctor, hospital to hospital... country to country. Kaufman RM et al. Ann Intern Med, Nov 11, 2014: Open access article  As mentioned in one of our earlier entries two important trials (Hermanns et al. The Lancet and Caesar et al. NEJM) have indicated that early parenteral nutrition (PN) might actually be harmful and a late PN strategy should be the standard of care. Despite this the debate has continued and remains highly controversial. This is nicely reflected in recent guidelines regarding the timing of supplemental PN. While the European Society for Clinical Nutrition and Metabolism (ESPEN) recommends PN within 24-48h in patients who are expected to be intolerant to enteral nutrition (EN), the American Society for Parenteral and Enteral Nutrition (ASPEN) recommends postponing the initiation of PN until day 8 after ICU admission. In order to clarify things a little Bost et al. have now published a systematic review on the timing of parenteral nutrition in critical care (Open access). From 3520 initially screened articles only four randomised controlled trials and two prospective observational studies remained after critical appraisal. "In conclusion it seems to be reasonable to assume that in critically ill patients, when full enteral support is contraindicated or fails to reach caloric targets, there are no clinically relevant benefits of early PN compared to late PN with respect to morbidity or mortality end points. Considering that infectious morbidity and resolution of organ failure may be negatively affected through mechanisms not yet clearly understood and acquisition costs of parenteral nutrition are higher, the early administration of parenteral nutrition cannot be recommended." The Review in one short sentence: Early PN has no advantage over late PN, but might be actually harmful. Bost et al. Annals of Intensive Care 2014, 4:31 Read more: ESPEN Guidelines on parenteral nutrition, 2009 A.S.P.E.N. Guidelines for the provision and assessment of nutrition support therapy in the adult critically ill patient BIJC.org on: Starting early PN weakens the critically ill  One of the key elements within antimicrobial stewardship and treatment paradigms for serious sepsis is de-escalation of antibiotic treatment. Antibiotic de-escalation describes a mechanism whereby the provision of effective initial antibiotic treatment is achieved while avoiding unnecessary antibiotic use that would promote the development of resistance. Basically it means that empirical antibiotics are stopped stopped or reduced in number and/or narrowed in spectrum on the basis of culture results. The recommendation to do so is mainly supported by observational studies only. This makes the publication of Leone at al. in Intensive Care Medicine of last month remarkable as they are the first to present a multi-center, non-blinded, randomised non-inferiority trial on de-escalation versus continuation of empirical antimicrobial treatment in severe Sepsis. After screening they included 120 patients of which 60 were assigned to continuation of appropriate empirical antimicrobial treatment and the other 60 to de-escalation. As a result de-escalation did not improve ICU length of stay (primary endpoint). De-escalation was associated though with an increased number of super infections (secondary endpoint), but did not affect mortality (secondary endpoint). Despite the studies limitations (relatively small population, lack of blind treatment and some other minor details) these results come surprisingly and leave us with the conclusion: De-escalation of antimicrobial treatment might remain standard of care at this stage but should be questioned if this is true in general. Leone M et al. Intensive Care Med (2014) 40:1399–1408 Masterton RG, Crit Care Clin (2011), Vol 27  Several studies in the past have looked into the topic of red blood cell (RBC) transfusions in the ICU and each one of them supports a rather restrictive approach in the ICU. Still though various guidelines around the world vary due to the lack of evidence (see below). This october the New England Journal of Medicine (unnoticeably and slowly transforming into a critical care journal ;) published a large multi-centered, partially blinded trial that randomised septic patients in intensive care units to receive RBC at a threshold of 70g/L or 90g/L. The primary outcome was mortality after 90 days. A total of 998 patients finally underwent randomisation and as a result there was no significant difference in mortality after 90 days. Also there were no statistically significant differences in all secondary endpoints like use of life supporting measures, ischemic events, and severe adverse reactions. This trial adds up to a list of studies showing that a liberal transfusion strategy is not beneficial for patients in critical care. This seems to be especially true for patients with sepsis. And not to forget: a considerable amount of packed RBC can be saved this way. A higher transfusion threshold of 90g/L in patients with sepsis is non-superior to a lower threshold of 70g/L. Get an insight into this topic yourself, here's the 'must read's about transfusions: The TRICC trial The CRIT study Sherwood M et al. JAMA. 2014 Feb 26;Vol 311, No.8 The FOCUS trial A short educational overview can be found here: http://lifeinthefastlane.com/education/ccc/blood-transfusion-in-icu/ Clinical Practice Guidelines from the AABB 2012: Ann Inten Med. 2012;157:49-58 Clinical Practice Guidelines 2009: Red blood cell transfusion in adult trauma and critical care, Crit Care Med 2009  Beeing involved in this multicenter, double-blind trial myself I am honoured to announce the Harp-2's publication in the New England Journal of Medicine. Congratulations not only to John Laffey but also to Michael Scully who kept Galway going in the recruitment of patients for this trial. In the past several publications have shown a beneficial effect of statins in the process of lung inflammation and injury. So far though none of these studies was designed or powered to show the effect of statins on clinical outcomes. The aim of this trial was therefore to see whether 80mg of daily simvastatin would improve clinical outcomes in patients with ARDS. For this study 40 intensive care units in Ireland and the United Kingdom (the ICU at the Galway University Hospital being one of them) randomised a total of 540 patients to receive either simvastatin 80mg OD or placebo. Primary outcome was defined as the number of ventilator-free days to day 28. Secondary outcomes included: change in the oxygenation index and the SOFA score up to day 28, the number of days free of non pulmonary organ failure to day 28, death from any cause within 28 days, death before discharge from ICU or the hospital, and safety. As a result there were no differences in any of the outcomes mentioned. As the authors mention in the discussion themselves: The use of simvastatin in the management of ARDS cannot be supported. McAuley et al. September 30, 2014DOI: 10.1056/NEJMoa1403285 Enteral Naloxon Safely Prevents Opioid Induced Constipation... Now also Confirmed in the NEJM26/9/2014
 It's not really the biggest news, but it's in the NEJM and it lines up with several publications looking at preventing opioid induced constipation with enteral naloxone. The article is remarkable in that sort of way as it represents a quite big double blinded study, while many publications before had a rather descriptive character. It has been shown before, that enteral naloxone helps reduce constipation when administered in conjunction with oxycodone. Interestingly enteral administration of naloxone blocks opioid action at the intestinal receptor level, but has a low systemic bioavailability due to marked hepatic first pass metabolism. In this study the investigators used naloxegol which is a PEGylated form of naloxone. This means that polyethylene glycol (PEG) polymer chains were covalently attached to the naloxone molecule. Chey WD et al. now basically present 2 double blinded studies including 625 patients in one and 700 patients in the other. Outpatients with non-cancer pain who were already getting opioids and experienced constipation were randomly assigned to receive either enteral naloxone (12.5mg or 25mg) or placebo. Primary endpoint was clinical response after twelve weeks defined as more than 3 bowel motions per week or an increase in bowel motions. Result: Enteral naloxone significantly reduced time to to the first post dose spontaneous bowel motion and increased the frequency of bowel motions. It is important to note that enteral naloxone did not reduce opioid mediated analgesia. Enteral naloxone should be considered in prevention or treatment of opioid induced constipation, also in the ICU. Chey WD et al. N Engl J Med. (2014) Webster et al. Aliment Pharmacol Ther 2014 Oct;40(7):771-9 This might also be of interest: Meissner et al. Crit Care Med. 2003 Mar;31(3):776-80, Enteral naloxone reduces gastric tube reflux and frequency of pneumonia in critical care patients during opioid analgesia.  Anaemia is a very common finding in critically ill patients and the idea to Supplement IV iron in these patients sounds tempting. Intravenous iron preparations are licensed for patients with iron deficiency anaemia when oral iron preparations are ineffective or contraindicated. The question is whether IV iron is also helpful in the critically ill. Pieracci et al. have looked at this question more precisely and published their results in Critical Care Medicine. In their multicentre, randomized, single-blind, placebo-controlled study they enrolled a total of 150 critically ill trauma patients in which baseline iron markers were consistent with functional iron deficiency anaemia. They randomized patients to either receive iron sucrose 100mg IV or placebo three times a weeks for up to 2 weeks. They found that treatment with IV iron increased ferritin concentration significantly but had no effect on transferrin saturation, iron-deficient erythropoiesis, haemoglobin concentration or packed RBC transfusion requirement. In conclusion: IV iron supplementation in anaemic, critically ill trauma patients cannot be recommended. Pieracci F et al. Crit Care Med, September 2014 - Volume 42 - Issue 9 - p 2048–2057  After a pause and thanks to Kuno's attentiveness following article found it's way to our website: Although not being very popular in the world of intensive care Digoxin remained a component of our armament and was continued to be used for patients with atrial fibrillation. After its long era in medicine findings of the TREAT-AF are now about to bring this to a possible end. Turakhia et al. looked at over 122'000 patients with newly diagnosed, non valvular AF in the U.S. between 2004 to 2008. They specifically looked at the use of Digoxin and the occurrence of death. Residual confounding was assessed by sensitivity analysis. They found a cumulative higher mortality rate for patients treated with Digoxin, which persisted after multivariate adjustment, propensity matching and adjustment for drug adherence. The findings of this study are impressive and even led Harlan Krumholz, editor-in-chief of NEJM Journal Watch Cardiology, to the statement: 'It's time to pause on Digoxin until studies can assure that it's providing a net benefit to these patients'. Turakhia et al. JACC, Aug 19 2014; Volume 64, Issue 7 NEJM Journal Watch Cardiology  Ventilator associated pneumonia (VAP) is a problem in ICU around the world and methicillin-resistant Staphylococcus aureus (MRSA) is the most common multi-drug resistant pathogen to deal with. Current guidelines mostly recommend vancomycin as a first line treatment and linezolid as an alternative, considering both drugs at a similar level of efficacy. The question remained whether linezolid might be superior to vancomycin. So far only one prospective, randomised, double-blind trial looked at this question and found a better success rate for linezolid, which was not statistically significant though. To look at this issue the IMPACT-HAP investigators (Improving Medicine through Pathway Assessment of Critical Therapy in Hospital Acquired Pneumonia) performed a multicenter, retrospective, observational study on 188 patients in 5 hospitals of the U.S. They found a significantly higher success rate with linezolid compared to vancomycin in the means of improvement or resolution of the signs and symptoms of VAP (primary endpoint). The study did not identify any difference though between linezolid- and vancomycin-treated patients in regards to mortality, development of thrombocytopenia, anaemia, or nephrotoxicity, days of mechanical ventilation or length of stay ion ICU or the hospital itself (secondary outcomes). Looking into the trial there appear to be several confounding reasons why patients treated with linezolid had better clinical success rate like less severity of sickness in linezolid patients, possible suboptimal vancomycin through levels etc. Overall there seems no good reasons at this stage to change current guidelines. Wunderink RG et al. Linezolid in methicillin-resistant Staphylococcus aureus nosocomial pneumonia, Clin Infect Dis; 2012, 54:621–629 Peyrani P et al. Crit Care 2014; 18:R118 doi:10.1186/cc13914  Thoracocentesis is a procedure often used to further determine pleural effusions. This commonly performed procedure may lead to complications including the development of a pneumothorax. Wilcox et al. performed a systematic review in JAMA of current literature to answer following two questions: 1. What are the most accurate diagnostic indicators to diagnose an exsudate? and 2. What are the most common adverse side effects and factors affecting them? 48 studies were included to answer the first question, while 37 studies were used to approach question number 2. In conclusion: - Light's criteria, pleural fluid cholesterol (<55 mg/dl) and pleural fluid LDH (>200 U/L) levels, and the pleural fluid cholesterol to serum cholesterol ratio (> 0.3) are the best diagnostic indicators for pleural exsudates - Pneumothorax was the most common complication of thoracocetesis (incidence 6%). Chest tube placement was needed in 2% of all procedures. And most impressive: Ultrasound skin marking by a radiologist or ultrasound-guided thoracocentesis were not associated with a decrease in pneumothorax events. ... would you abandon the ultrasound? Wilcox et al. JAMA, June 18, 2014, Vol 311, No. 23 |
Search
|

 RSS Feed
RSS Feed


